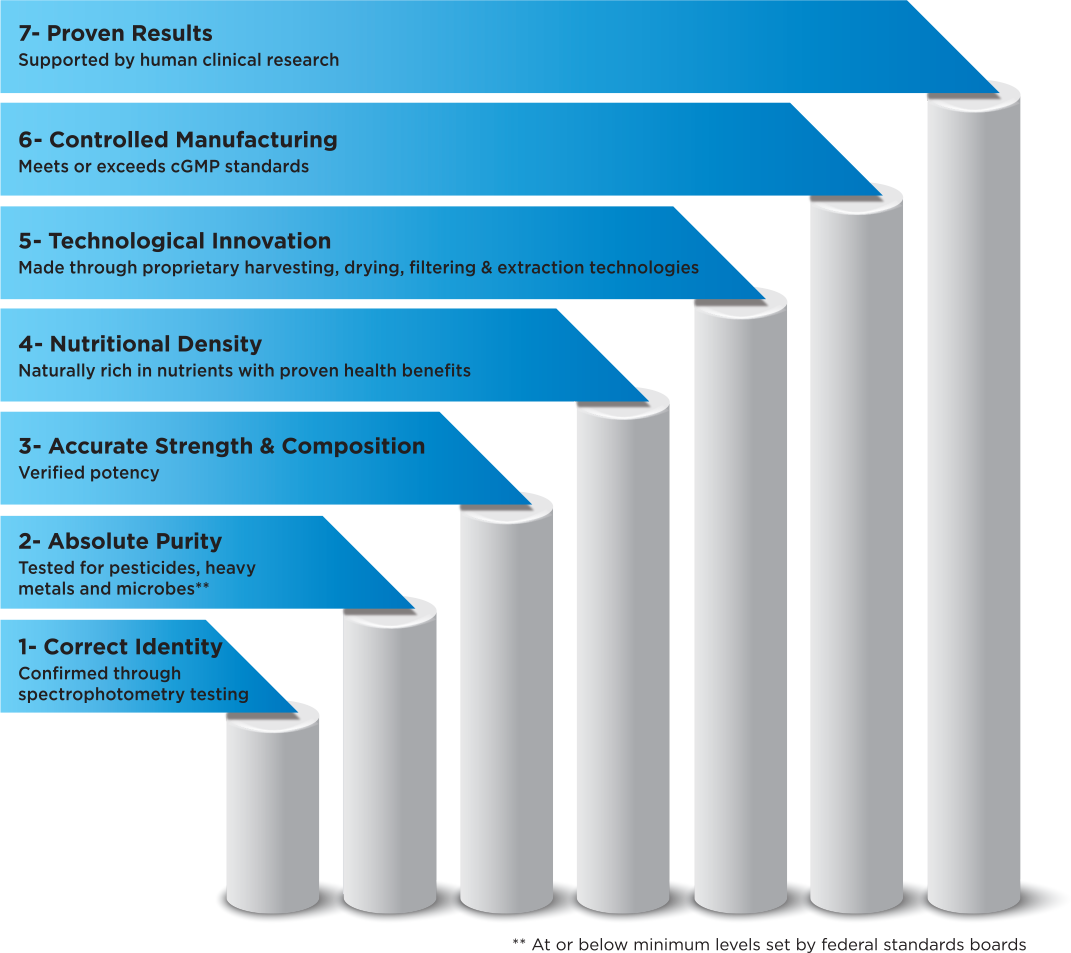
 Quality is a concern for any manufacturer of raw materials for use in nutraceutical and pharmaceutical applications. Therefore, it is of the utmost importance to choose a raw material supplier that has impeccable quality controls in place. Desert Lake Technologies adheres to a strict 7-point quality program that controls the safety, purity and potency of each product we manufacture.
Quality is a concern for any manufacturer of raw materials for use in nutraceutical and pharmaceutical applications. Therefore, it is of the utmost importance to choose a raw material supplier that has impeccable quality controls in place. Desert Lake Technologies adheres to a strict 7-point quality program that controls the safety, purity and potency of each product we manufacture.
Materials
Whether we wild-harvest it ourselves or purchase it from thoroughly vetted suppliers, Desert Lake Technologies's microalgae source material is grown and harvested under the strictest of quality guidelines and procedures. Before getting stamped with the Desert Lake Technologies seal of approval, each batch of material must meet strict product specification parameters and pass a painstaking regimen of identity testing, purity testing, and comparative test result analysis.
Processes
Desert Lake Technologies's in-process controls and Enterprise Resource Planning (ERP) System allow for complete traceability of our products. Selective harvesting, in-process testing and analysis, proprietary technologies, and on-site Quality Control personnel ensure the purity of our products during each step of the manufacturing process. Our in-house team of engineering, design, maintenance, and fabricating experts build, control, and maintain our state-of-the-art equipment.
Quality Management
Our quality team is comprised of industry experts who guide every aspect of our business affecting the quality of our finished products, including harvest, procurement, production, facilities, testing, and internal and vendor audits. In addition, this team oversees all regulatory affairs, ensuring complete compliance with regulations issued by FDA, OSHA, USDA, and ODA.
Testing
Desert Lake Technologies tests all of our source and finished materials for aerobic bacteria, yeast and mold, applicable pathogenic microbes, heavy metals, pesticides, and impurities. We provide a batch-specific Certificate of Analysis with each sale of product guaranteeing its safety and purity.
Certifications
- Oregon Tilth Certified Organic Processor and Harvester (an NOP approved program)
- Halal
- Kosher and Pareve
- GRAS (Cyactiv: FDA #GRN 000424)

 Quality is a concern for any manufacturer of raw materials for use in nutraceutical and pharmaceutical applications. Therefore, it is of the utmost importance to choose a raw material supplier that has impeccable quality controls in place. Desert Lake Technologies adheres to a strict 7-point quality program that controls the safety, purity and potency of each product we manufacture.
Quality is a concern for any manufacturer of raw materials for use in nutraceutical and pharmaceutical applications. Therefore, it is of the utmost importance to choose a raw material supplier that has impeccable quality controls in place. Desert Lake Technologies adheres to a strict 7-point quality program that controls the safety, purity and potency of each product we manufacture.




